Welcome to today’s exploration into one of the intriguing questions in the field of electrochemistry: do we actually need a potentiostat in modern applications? This question touches upon both historical developments and contemporary innovations that continue to shape scientific research and commercialization.
The Origins of Potentiostats
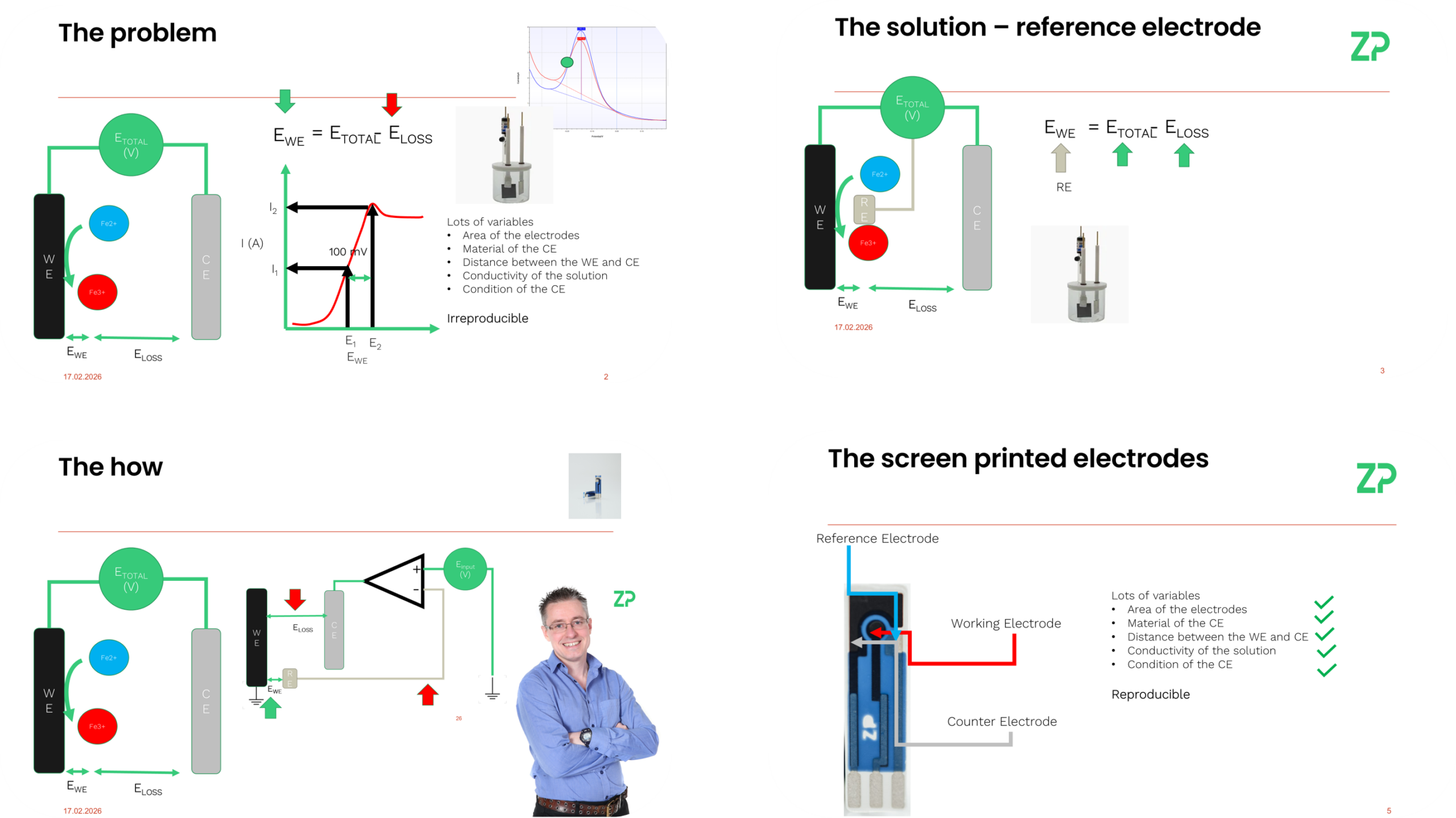
Potentiostats were developed during the early days of electrochemical science to address significant reproducibility issues. Electrochemists working with classical cell setups encountered challenges due to variable potentials when conducting experiments such as oxidizing ferrocyanide to ferricyanide. The non-linear nature of electrochemistry meant that even small changes in applied voltage could lead to considerable variations in current, making it difficult to achieve consistent results across different laboratories.
The problem was further compounded by numerous variables such as the area and material of electrodes, their distance from each other, and the conductivity of solutions—factors that differed widely between labs. To combat this, scientists introduced reference electrodes, which helped stabilize the potential near working electrodes when combined with potentiostat circuits. These circuits allowed for more precise control over applied voltages through feedback mechanisms.
The Role of Screen Printed Electrodes (SPEs)
One significant advancement that addresses many challenges once requiring potentiostats is the development of screen printed electrodes (SPEs). These devices integrate a working electrode, reference electrode, and counter electrode into a single, reproducible unit. By manufacturing these components with fixed parameters—such as electrode area, material type, and distance between components—many variables that previously affected reproducibility are now controlled.
This standardization has profound implications for both research and commercial applications. For instance, in the field of glucose monitoring, many devices no longer rely on traditional potentiostats to achieve consistent results. Instead, they use SPEs that come with built-in consistency and reliability.
Commercialization and Beyond
In the realm of commercialization, the necessity of a potentiostat is often re-evaluated. While essential for R&D and precise laboratory work, many commercial applications find alternatives that simplify processes without compromising performance. This shift raises an important question: are traditional three-electrode systems and specific reference electrodes like silver chloride truly necessary?
Looking Ahead
In upcoming articles, I will delve deeper into whether elements such as the classic three-electrode system or specific types of reference electrodes remain essential in today’s electrochemical landscape. We’ll explore how modern innovations continue to redefine what is possible, pushing the boundaries of both research and commercial applications.
If you have any questions or insights about electrochemical setups or the role of SPEs in current technology, feel free to reach out! Let's continue this fascinating journey together as we explore the ever-evolving world of electrochemistry.