Distinguishing Active HIV Infection from Vaccine-Induced Seropositivity Using Electrochemistry
A major challenge in HIV vaccine trials is a phenomenon known as vaccine-induced seropositivity (VISP). Many experimental HIV vaccines trigger the production of antibodies that look identical to those produced during real HIV infection when measured by standard diagnostic tests. As a result, vaccinated but uninfected trial participants can test “HIV-positive” using routine screening — creating serious clinical, psychological, and logistical complications.
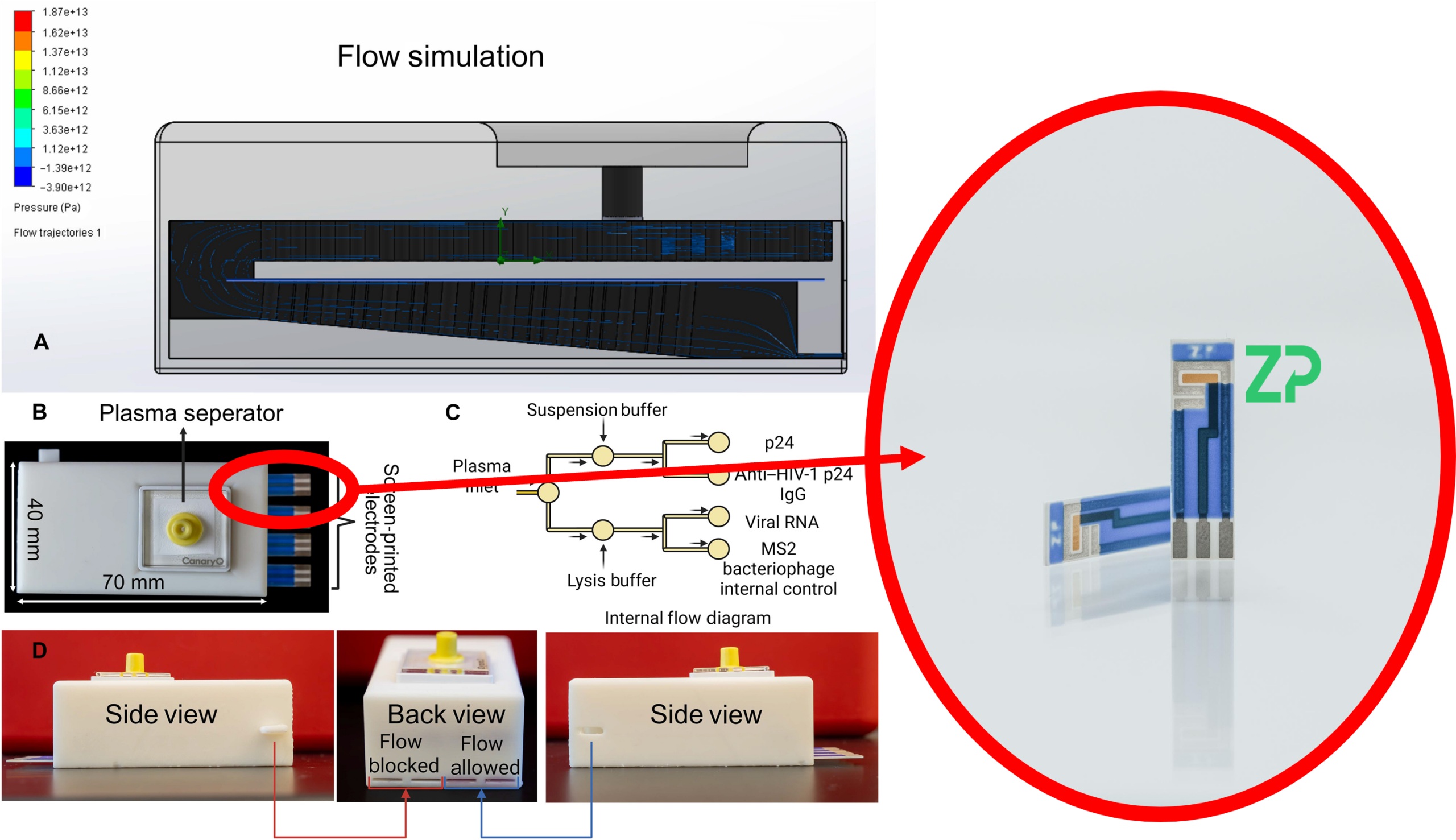
To solve this problem, researchers recently developed a rapid electrochemical companion diagnostic capable of clearly distinguishing true, active HIV-1 infection from vaccine-induced immune responses. A critical component of this technology was the use of Zimmer & Peacock (ZP) 303 Gold screen-printed electrodes (SPEs) as the core sensing platform.
This work highlights how commercial electrochemical sensor platforms can directly support next-generation diagnostics at the point of care.
The Diagnostic Challenge of VISP
HIV vaccines are designed to stimulate strong antibody responses. Unfortunately, those same antibodies are exactly what conventional HIV tests detect. This creates a scenario where:
A vaccinated individual may appear HIV-positive
Clinical follow-up becomes complicated
Trial participants face unnecessary stigma and anxiety
Long-term medical records can be affected
What is truly needed is a companion diagnostic — a complementary test that can determine whether a positive HIV antibody result reflects true viral infection or merely vaccine exposure.
The Electrochemical Solution
The research team developed a compact, electrochemical biosensing device that combines:
A 3D-printed microfluidic cartridge
Multiplexed biochemical probes
Screen-printed gold electrodes
A portable electrochemical reader
Instead of relying on a single antibody signal, the test simultaneously measures multiple biomarkers associated with:
Active viral infection
Vaccine-induced immune responses
By analyzing these combined electrical signals, the system can reliably differentiate between true HIV infection and VISP — delivering results in just minutes.
Why Screen-Printed Electrodes Were Essential
At the heart of the device is the Zimmer & Peacock 303 Gold screen-printed electrode, a commercially manufactured, research-grade electrochemical sensor featuring:
A gold working electrode
An integrated Ag/AgCl reference electrode
A compact 7 mm ceramic substrate
High reproducibility from batch to batch
These electrodes were chemically functionalized with:
HIV antigens
Antibodies
DNA probes tagged with electrochemical reporters
Once functionalized, the electrodes convert molecular binding events into electrical signals that can be rapidly measured.
Why Gold Matters
Gold is the preferred surface for many biosensors because it allows:
Stable thiol-based self-assembled monolayers (SAMs)
Highly controlled biomolecule attachment
Excellent electrochemical stability
High signal-to-noise performance
These properties make gold ideal for hosting complex assay chemistries involving proteins, oligonucleotides, and redox reporters. The ZP 303 Gold electrode provided exactly the surface chemistry needed for the researchers’ multiplexed detection strategy.
Key Advantages of Using Zimmer & Peacock 303 Gold SPEs
Using off-the-shelf ZP electrodes delivered several major advantages to the project:
1. Reproducibility
Each electrode is manufactured with consistent geometry, gold surface area, and reference electrode behavior — essential for clinical assay reliability.
2. Rapid Development
The team did not need to fabricate electrodes in-house, allowing them to move quickly from concept to functional prototype.
3. Integrated Reference Electrode
The built-in Ag/AgCl reference simplified the cartridge design and eliminated bulky external electrodes.
4. Point-of-Care Compatibility
The compact form factor is ideal for portable, battery-powered electrochemical readers — critical for decentralized testing environments.
5. Scalable Manufacturing
Because the electrodes are already mass-manufactured, the pathway from lab prototype to scaled clinical production is dramatically simplified.
Clinical Performance
In testing on over 100 human blood samples, the electrochemical assay demonstrated high sensitivity and specificity for distinguishing:
Active HIV-1 infection
Vaccine-induced antibody responses
Results were delivered in approximately five minutes, making the platform suitable for real-world clinical workflows, trial participant screening, and point-of-care diagnostics.
What This Means for Diagnostics and Biosensing
This work demonstrates an important shift in diagnostic development:
Commercial electrochemical sensor platforms like Zimmer & Peacock’s SPEs are no longer just research tools — they are now foundational components of clinical diagnostic devices.
Multiplexed electrochemistry enables smarter interpretation of immune responses, not just simple “positive/negative” results.
Rapid companion diagnostics can significantly accelerate vaccine trials, clinical decision-making, and patient confidence.
Conclusion
The ability to distinguish true HIV infection from vaccine-induced seropositivity represents a major breakthrough for HIV vaccine trials and participant care. By combining smart bioassay design, portable electrochemical instrumentation, and robust Zimmer & Peacock 303 Gold screen-printed electrodes, the researchers created a diagnostic platform that is:
Rapid
Highly specific
Clinically practical
And scalable for real-world deployment
This work is a powerful example of how screen-printed electrochemistry is driving the next generation of point-of-care diagnostics.