Introduction
Electrochemistry is fundamentally an interfacial science. Every time an electrode is polarised in solution, processes occur not only through redox reactions of analytes, but also through the reorganisation of ions at the electrode–electrolyte interface. These interfacial phenomena give rise to what is known as the electrical double layer and the associated charging current. Although rarely discussed in day‑to‑day applied electroanalysis, interfacial electrochemistry plays a critical role in how signals are formed, distorted, and ultimately filtered by modern instrumentation. Understanding it provides valuable insight into why techniques such as square‑wave or differential pulse voltammetry are so effective — and why modern potentiostats deliberately suppress parts of the signal. Supporting Video
Faradaic vs Non‑Faradaic Processes
At an electrode surface, two fundamentally different processes can occur:
✅ Faradaic Processes
These involve electron transfer reactions, such as the oxidation of ferrocyanide (Fe²⁺ → Fe³⁺). The resulting current is directly linked to the analyte concentration and is typically the signal of interest in electroanalytical measurements.
⚙️ Non‑Faradaic Processes
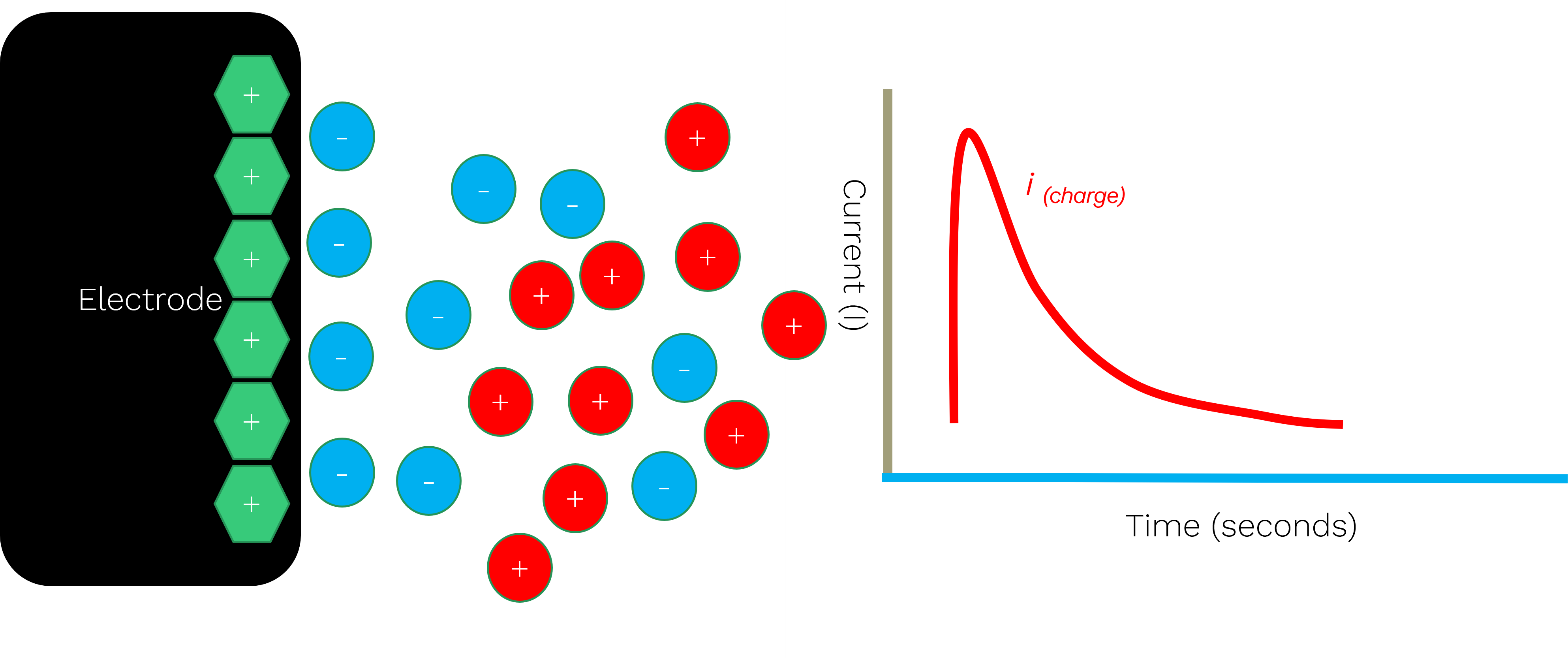
These arise from ion rearrangement near the electrode surface when a potential is applied. No electron transfer occurs; instead, ions in solution move to balance the electrode charge. This generates a transient current known as the charging current.
Both processes occur simultaneously whenever an electrode is polarised — but only one usually carries analytical value.
The Electrical Double Layer
When a potential is applied to an electrode:
- The electrode surface becomes charged
- Counter‑ions in solution migrate toward the surface
- A structured region of charge forms at the interface
This structure is referred to as the electrical double layer, and its formation is responsible for the non‑faradaic charging current observed immediately after a potential step.
Classical Models of the Electrical Double Layer
Over nearly two centuries, several models have been proposed to describe this interfacial structure. While largely academic today, they remain useful for conceptual understanding.
🔬 Helmholtz Model (1850s)
The earliest model treats the interface as a rigid, ordered layer of ions aligned directly against the electrode surface.
- Ions are fixed in position
- No thermal motion is considered
- The interface resembles a simple capacitor
Limitation: Over‑simplified and unrealistic for liquid electrolytes.
🔬 Gouy–Chapman Model (circa 1910)
This model introduced the idea of thermal motion and treats ions as forming a diffuse cloud rather than a rigid layer.
- Accounts for ion mobility
- Predicts a gradual decay of charge away from the electrode
- More realistic for aqueous systems
Limitation: Treats ions as point charges and ignores finite ion size.
🔬 Stern Model (1924)
The Stern model combines the strengths of both earlier approaches:
- A compact, structured Stern layer close to the electrode
- A surrounding diffuse layer extending into solution
This hybrid model correlates far better with experimental data and is widely accepted as “good enough” for most practical purposes.
Why Charging Current Matters — and Why It’s Often Ignored
Whenever a potential is stepped or swept, the electrical double layer must rearrange. This produces a sharp, transient current spike that:
- Is non‑faradaic
- Does not relate to analyte concentration
- Can obscure meaningful electrochemical signals
In practical electroanalysis, this current is considered noise, not information.
How Modern Instrumentation Deals with It
Most modern potentiostats — including Zimmer & Peacock systems — deliberately filter out charging current using time and technique selection.
📌 Time‑Based Filtering
During linear sweep voltammetry, the initial current spike following each potential step is ignored. Measurement occurs only after the double layer has stabilised.
📌 Technique‑Based Suppression
Methods such as:
- Differential Pulse Voltammetry (DPV)
- Square‑Wave Voltammetry (SWV)
use forward/reverse scans and signal subtraction to minimise non‑faradaic contributions, dramatically improving signal‑to‑noise ratios.
This is why interfacial electrochemistry, while always present, is rarely visible in final datasets.
Practical Context: Electrodes and Potentiostats
In applied sensing — for example using screen‑printed electrodes — electrical double layer effects occur continuously, whether measuring caffeine, capsaicin, or ionic species.
Modern systems such as the SenseItAll Generation 4 potentiostat are designed with these realities in mind, combining:
- Fast electronics
- Intelligent timing
- Advanced measurement techniques
to ensure users focus on chemistry that matters, not interfacial artefacts.
🔗 Screen‑printed electrodes:
https://shop.zimmerpeacock.com/en-gb/collections/bare-electrodes
🔗 SenseItAll Gen 4 potentiostat:
https://shop.zimmerpeacock.com/en-gb/products/senseitall-sia-generation-4-device-only
Key Practical Takeaways
✅ Electrochemistry is inherently interfacial
✅ Charging current originates from electrical double layer formation
✅ Classical models explain why it exists, not how to measure it
✅ Modern techniques and instruments deliberately suppress it
✅ Good electroanalysis depends as much on instrumentation strategy as chemistry
Closing Thoughts
While interfacial electrochemistry is often treated as background theory, it quietly underpins every electrochemical measurement. Understanding it helps practitioners interpret data more confidently, select appropriate techniques, and appreciate why modern electrochemical systems behave the way they do.
If you’d like to discuss applications, instrumentation choices, or measurement strategies in more depth, the Zimmer & Peacock team is always happy to explore ideas and challenges collaboratively.
🔗 Get in touch:
https://www.zimmerpeacock.com/contact