Monitoring the partial pressure of carbon dioxide (pCO₂) is essential across a surprising range of applications—from bioreactors and blood gas analysis to even swimming pool monitoring. In this post, we’ll break down what pCCO₂ actually represents, why it matters, and how modern miniature sensors—such as screen‑printed electrodes—make these measurements possible.
Why Measure pCO₂?
pCO₂ refers to the partial pressure of carbon dioxide within a sample. In gas–liquid systems, this is straightforward: CO₂ in the gas phase equilibrates with CO₂ dissolved in the liquid. But even in systems without a true gas phase—like blood—the concept still applies because dissolved CO₂ behaves as if it were in equilibrium with a gas phase.
This makes pCO₂ a valuable parameter in:
Clinical blood analysis
Bioprocess monitoring
Environmental and water‑quality applications
The Sensors: Small but Powerful
The form factors used for pCCO₂ sensing are impressively compact. A typical screen‑printed electrode might measure only 7 mm × 26 mm—roughly an inch long and a third of an inch wide. Despite their size, these sensors can deliver high‑quality chemical measurements when paired with the right calibration and supporting sensors.
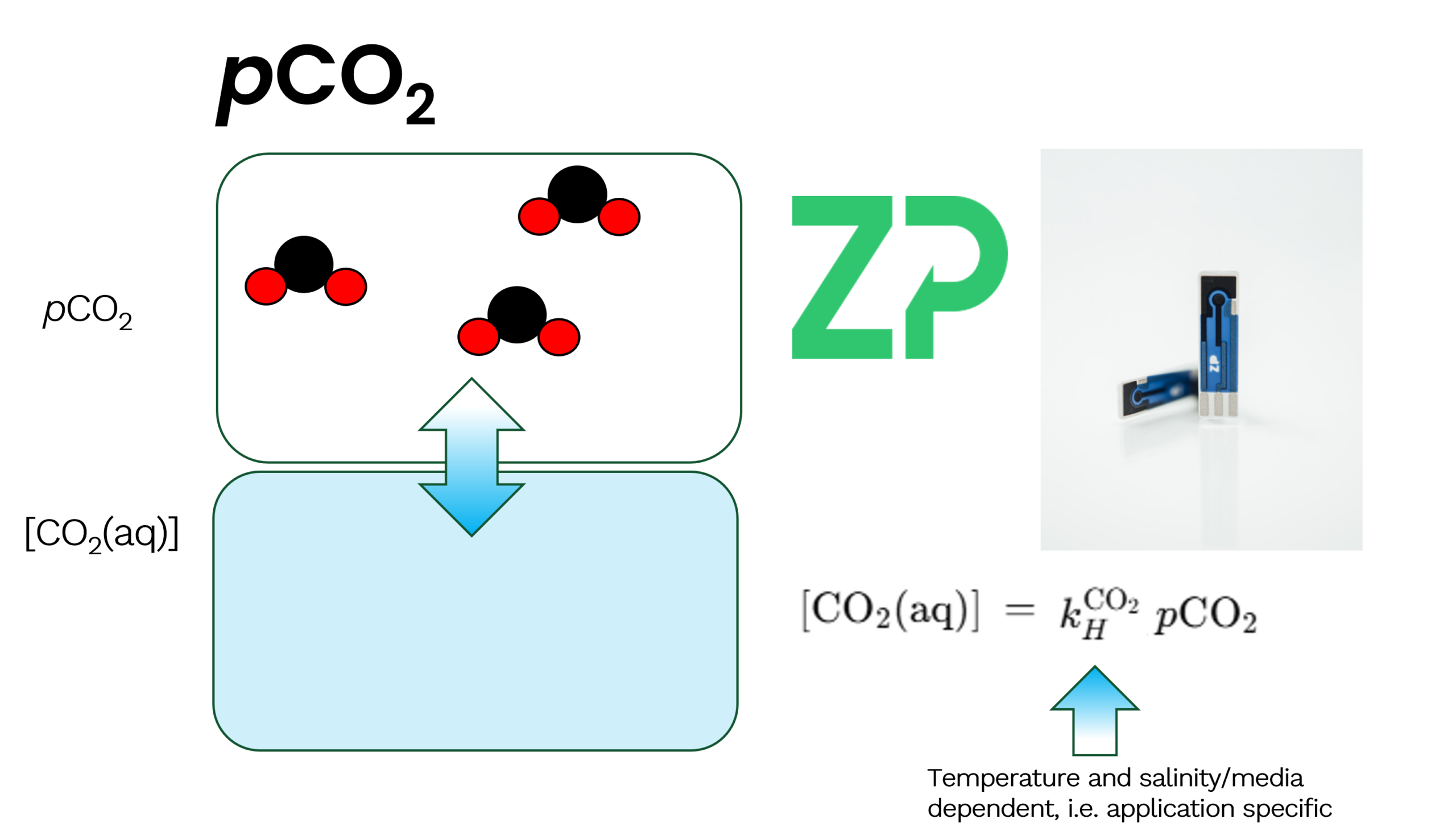
Henry’s Law: The Foundation of pCO₂ Measurement
At the heart of pCCO₂ measurement is Henry’s Law, which states:
[CO2]aq = K x pCO2
Where:
[CO₂(aq)] is the concentration of dissolved CO₂
K is Henry’s constant
pCO₂ is the partial pressure of CO₂
Importantly, K is not universal. It varies with:
Temperature
Salinity
The chemical composition of the medium
This means that blood, seawater, bioreactor broth, and swimming pool water all require their own calibration.
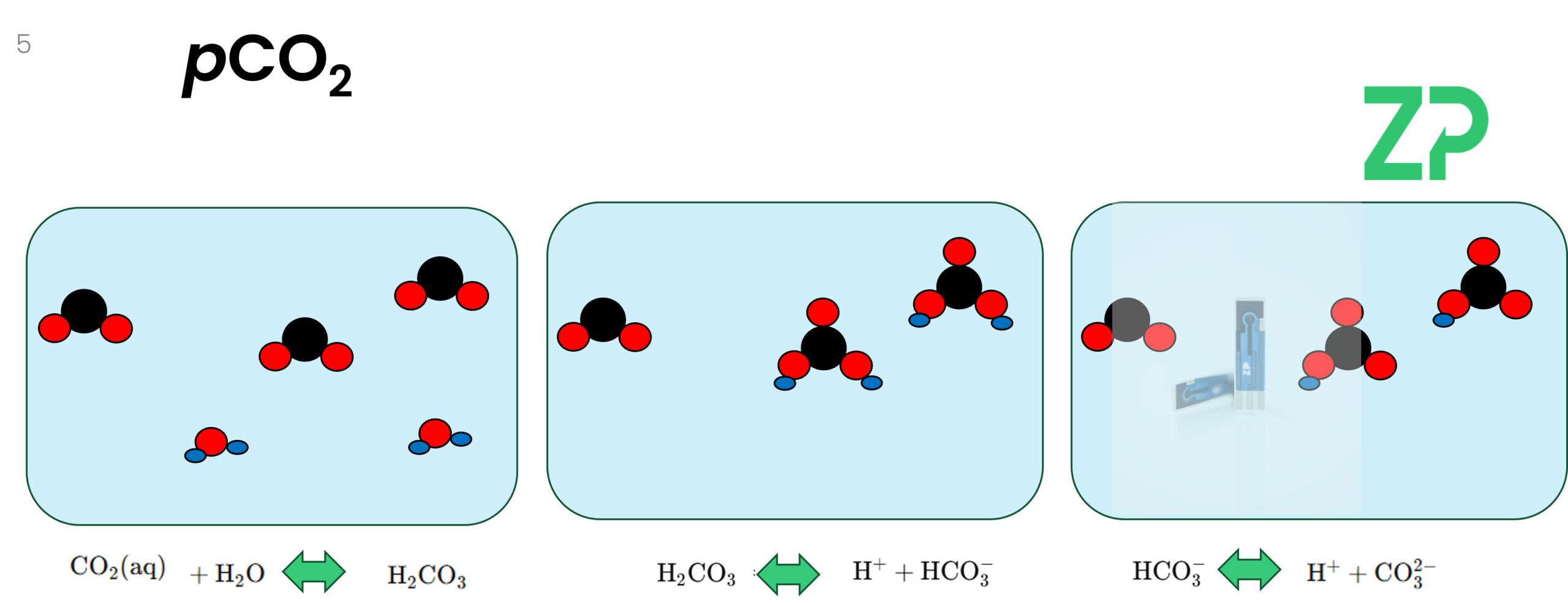
CO₂ Chemistry in Solution: More Than Just Dissolved Gas
Once CO₂ dissolves in water, it doesn’t stay as CO₂(aq). It reacts to form a series of related species:
CO₂ + H₂O → H₂CO₃ (carbonic acid)
H₂CO₃ ⇌ HCO₃⁻ + H⁺ (bicarbonate)
HCO₃⁻ ⇌ CO₃²⁻ + H⁺ (carbonate)
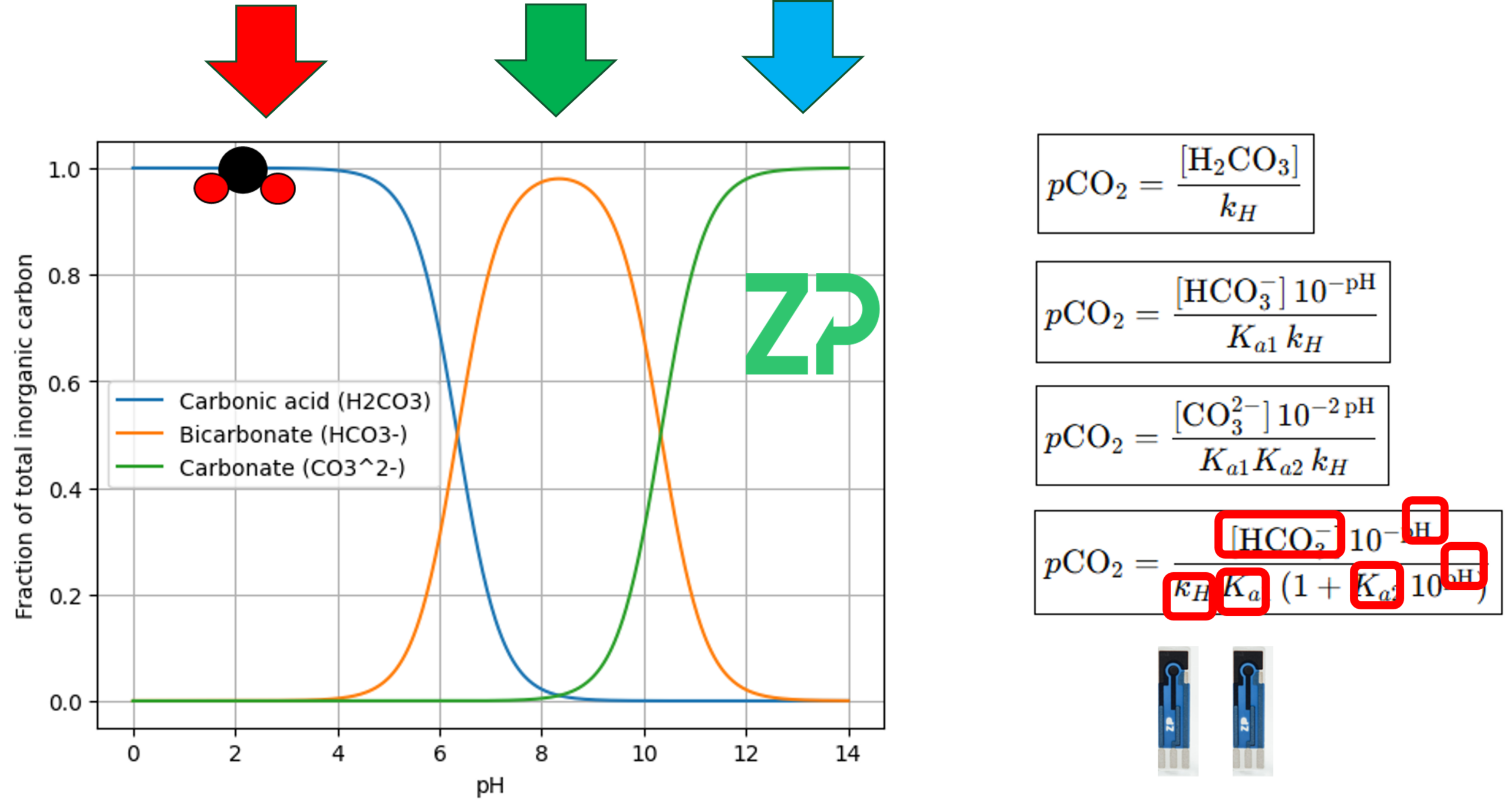
The dominant species depends heavily on pH:
Low pH: Mostly carbonic acid
pH 6–8: Mostly bicarbonate
High pH (>11): Mostly carbonate
This matters because ZP sensing technology measures bicarbonate, not CO₂ directly. By combining a bicarbonate sensor with a pH sensor, we can mathematically determine pCO₂.
How Blood Gas Analyzers Do It
Most blood gas analyzers don’t measure pCO₂ directly. Instead, they:
Measure bicarbonate
Measure pH
Apply known equilibrium constants
Calculate pCO₂
This same approach works for miniature electrochemical sensors.
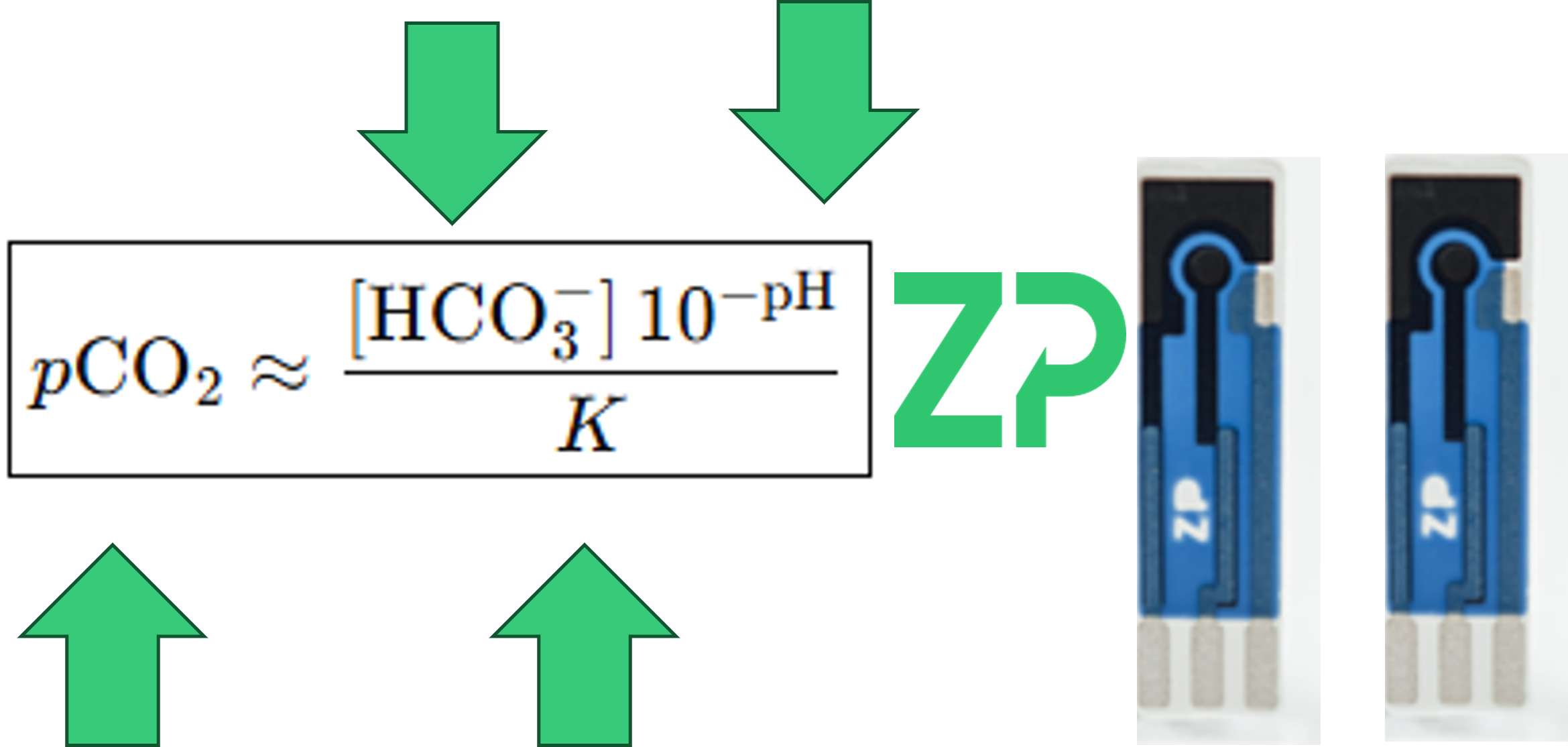
A Universal Equation for pCO₂
Because the relationships between CO₂, bicarbonate, and pH are well‑defined, we can use a universal equation to compute pCO₂ at any pH—provided we know:
Bicarbonate concentration (measured)
pH (measured)
K (determined through calibration for the specific medium)
Once calibrated, the system becomes robust and highly adaptable across different applications.
Bringing It All Together
To determine pCO₂ using small‑form sensors, you need only:
A bicarbonate sensor
A pH sensor
A calibration routine to determine the appropriate K value for your medium
With these components, even tiny screen‑printed electrodes can deliver accurate, application‑specific pCCO₂ measurements.
If you’d like help selecting or integrating pH and bicarbonate sensors for your application, feel free to reach out. These compact sensing platforms open up powerful possibilities for real‑time chemical monitoring.