Why Partial Pressure of Oxygen Matters Pₒ₂ describes the pressure that oxygen contributes within a mixture of gases. In physiology, it’s used to understand how much oxygen is available to tissues via the bloodstream. Clinicians rely on Pₒ₂ to assess respiratory function, oxygenation efficiency, and overall patient health. But here’s the important link: Pₒ₂ determines how much oxygen dissolves into a liquid, such as blood or water. And that dissolved oxygen is what electrochemical sensors actually measure.
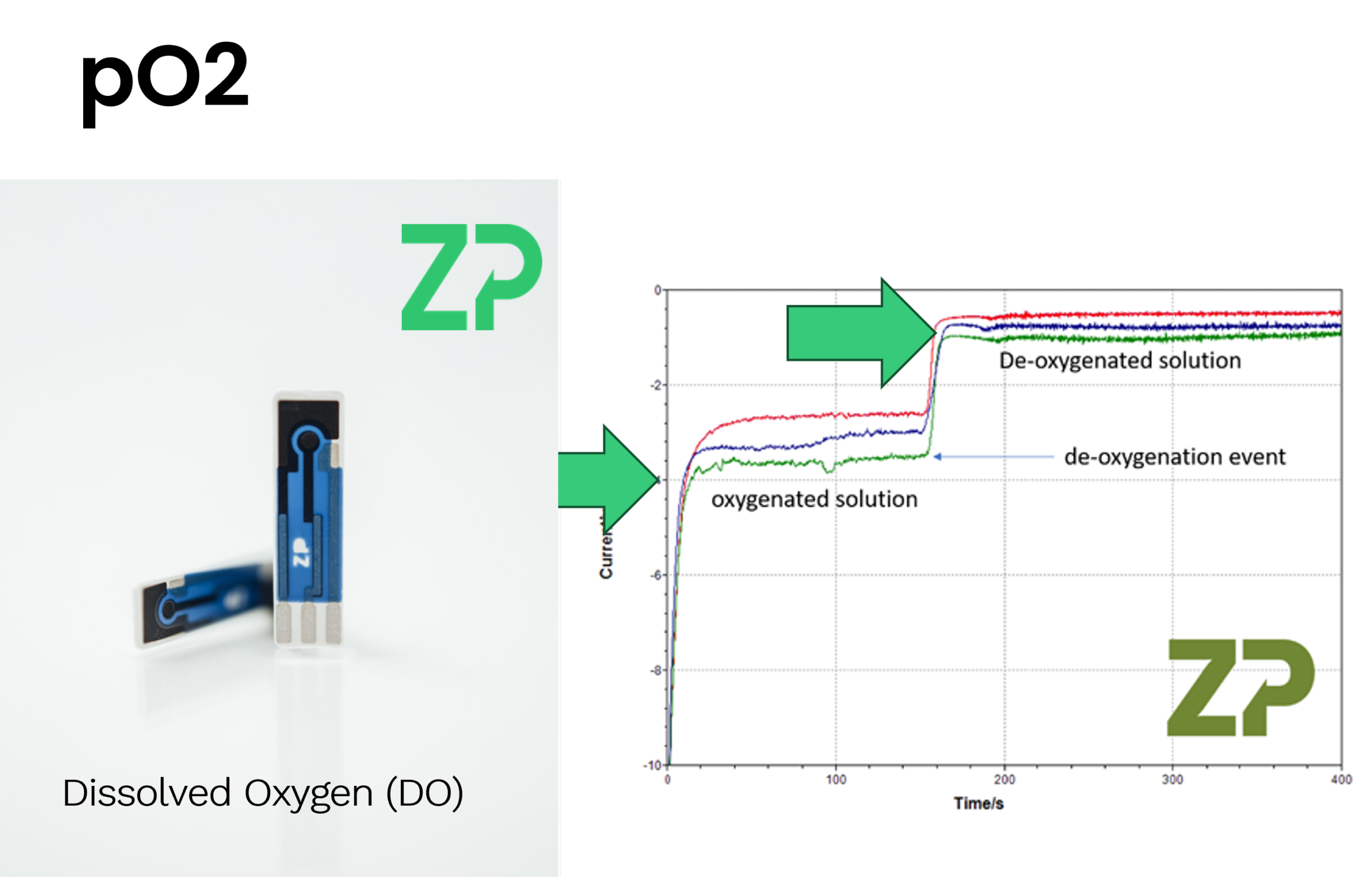
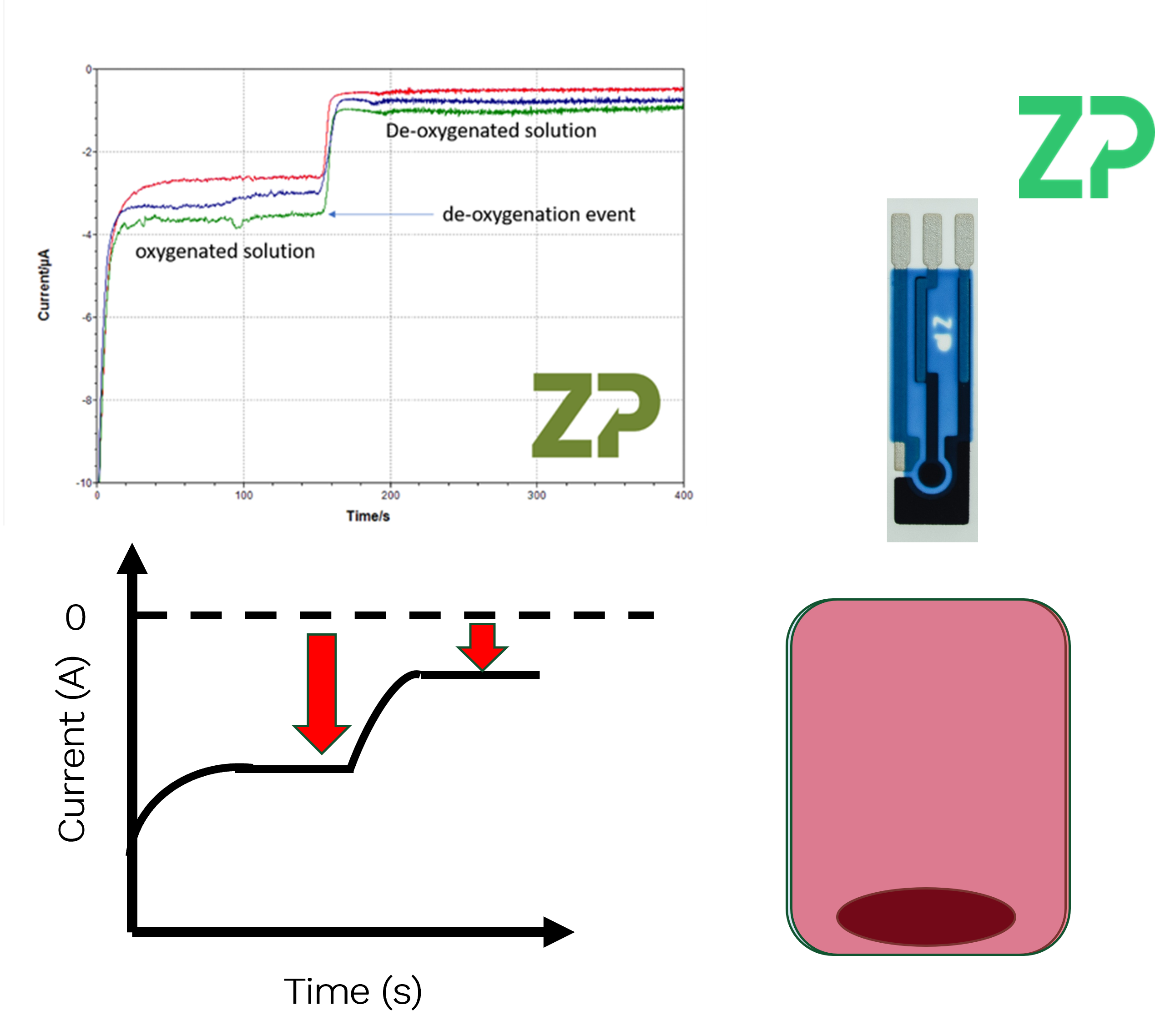
Measuring Dissolved Oxygen with Sensors At Zimmer & Peacock, we use electrochemical dissolved oxygen sensors to quantify oxygen levels in liquids. On our datasheets, you’ll see plots showing the sensor’s signal under oxygenated and deoxygenated conditions. At first glance, the magnitude of the signals can seem counterintuitive—the oxygenated signal may appear lower than the deoxygenated one—but this is simply due to how the current responds to oxygen reduction at the electrode surface. A Typical Demonstration When we demonstrate the sensor:
- We place it into a solution containing oxygen.
- The sensor initially shows a high current due to a phenomenon called charging current.
- The signal quickly stabilizes (plateaus), and this plateau corresponds to the dissolved oxygen concentration.
- If we add an oxygen scavenger, the oxygen level drops sharply.
- The sensor signal drops to a new, lower plateau—representing near-zero oxygen. The difference between the two plateaus is directly related to the oxygen concentration in the solution.
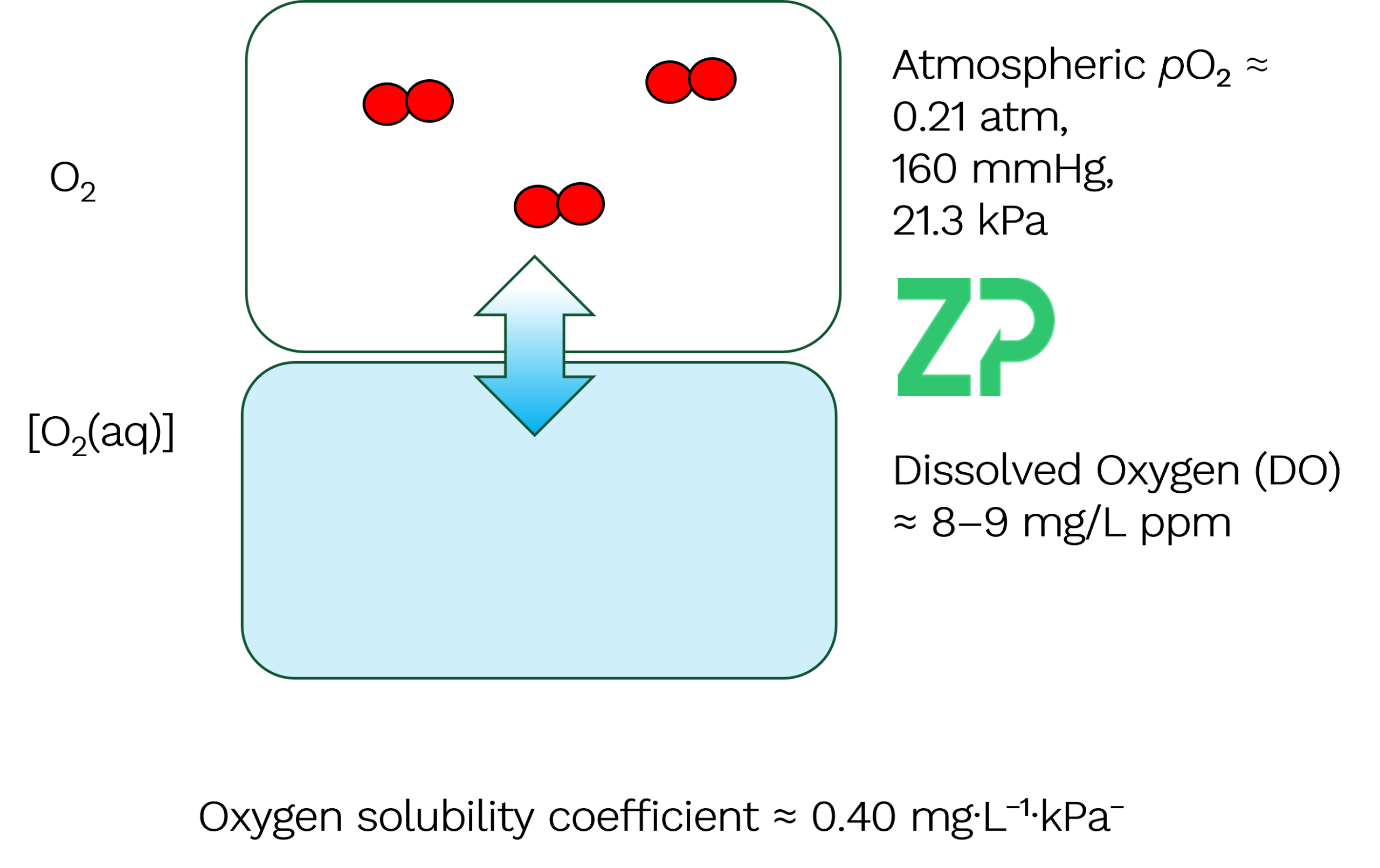
Connecting Dissolved Oxygen to Partial Pressure Now let’s connect this to Pₒ₂. At atmospheric conditions, oxygen makes up about 21% of the air. This corresponds to: • ~0.21 atmospheres • ~166 mmHg • ~21.3 kPa Under these conditions, water at room temperature typically contains 8–9 mg/L (or ppm) of dissolved oxygen. Henry’s Law If you have a chemistry background, you’ll recognize this relationship from Henry’s Law, which states:
Mathematically:
Where: • = dissolved oxygen concentration • = partial pressure of oxygen • = Henry’s constant (depends on gas, solvent, and temperature) Physiological Interpretation In physiology, we often express this relationship using a solubility coefficient instead of Henry’s constant. For oxygen in water, a typical solubility coefficient is: • 0.04 mg/L per kPa, or • 0.055 ppm per mmHg in physiological solutions
Example: Converting Dissolved Oxygen to Pₒ₂ Imagine we measure a dissolved oxygen concentration of 4.3 ppm in a flowing or pumped sample (such as blood or a physiological buffer). Using the solubility coefficient:
So a dissolved oxygen reading of 4.3 ppm corresponds to a Pₒ₂ of about 78 mmHg. This is exactly how dissolved oxygen sensors help us infer physiologically meaningful parameters.
Calibration and Practical Considerations To ensure accurate Pₒ₂ measurements: • Calibrate the sensor in the same medium you plan to measure (blood, buffer, water, etc.). • Remember that solubility coefficients vary between media. • Temperature matters—oxygen solubility decreases as temperature increases. • Always control or record temperature when converting between dissolved oxygen and Pₒ₂.
Final Thoughts Partial pressure of oxygen is a fundamental parameter in both clinical diagnostics and scientific research. By understanding the relationship between Pₒ₂ and dissolved oxygen—and by using well-calibrated sensors—you can obtain accurate, meaningful measurements in a wide range of applications. If you have any questions about Pₒ₂ measurement or dissolved oxygen sensing, feel free to reach out to us at Zimmer & Peacock. We’re always happy to help.