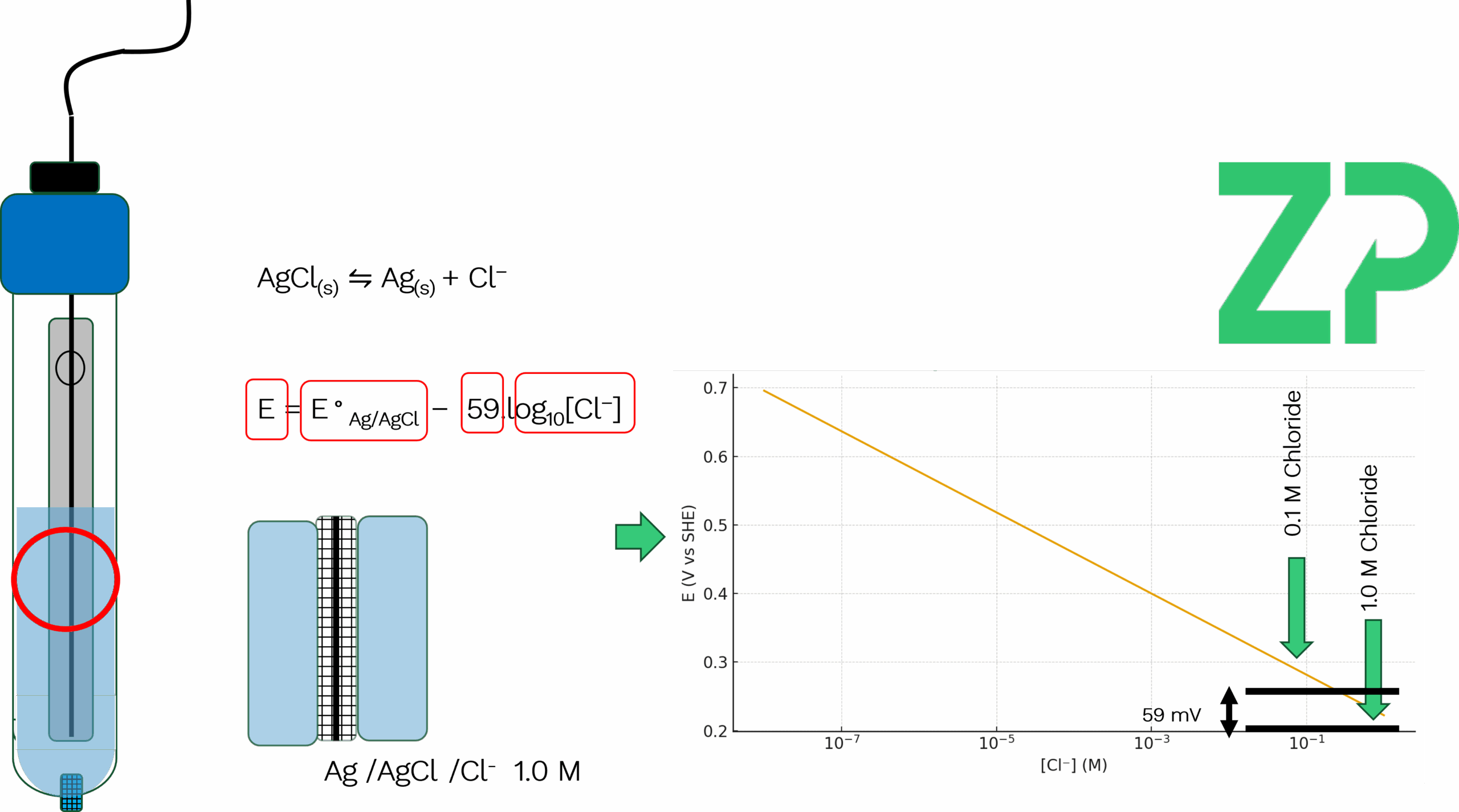
Understanding Pseudo Reference Electrodes: A Practical Guide Welcome to this deep dive into the world of reference electrodes—specifically, the distinction between true reference electrodes and pseudo reference electrodes. Whether you're working in a traditional electrochemistry lab or developing cutting-edge wearable sensors, understanding your reference electrode is critical to getting accurate and reproducible results. 🧪 What Is a True Reference Electrode? Let’s start with the classic: the silver/silver chloride (Ag/AgCl) reference electrode. If you've worked in a lab, you've likely seen one. It typically consists of: • A glass barrel or cylinder • A porous frit or ceramic plug at the bottom • A silver wire coated in silver chloride • A potassium chloride (KCl) solution, often 1.0 M • A cap to seal the system This setup ensures a stable and well-defined potential, governed by the Nernst equation:
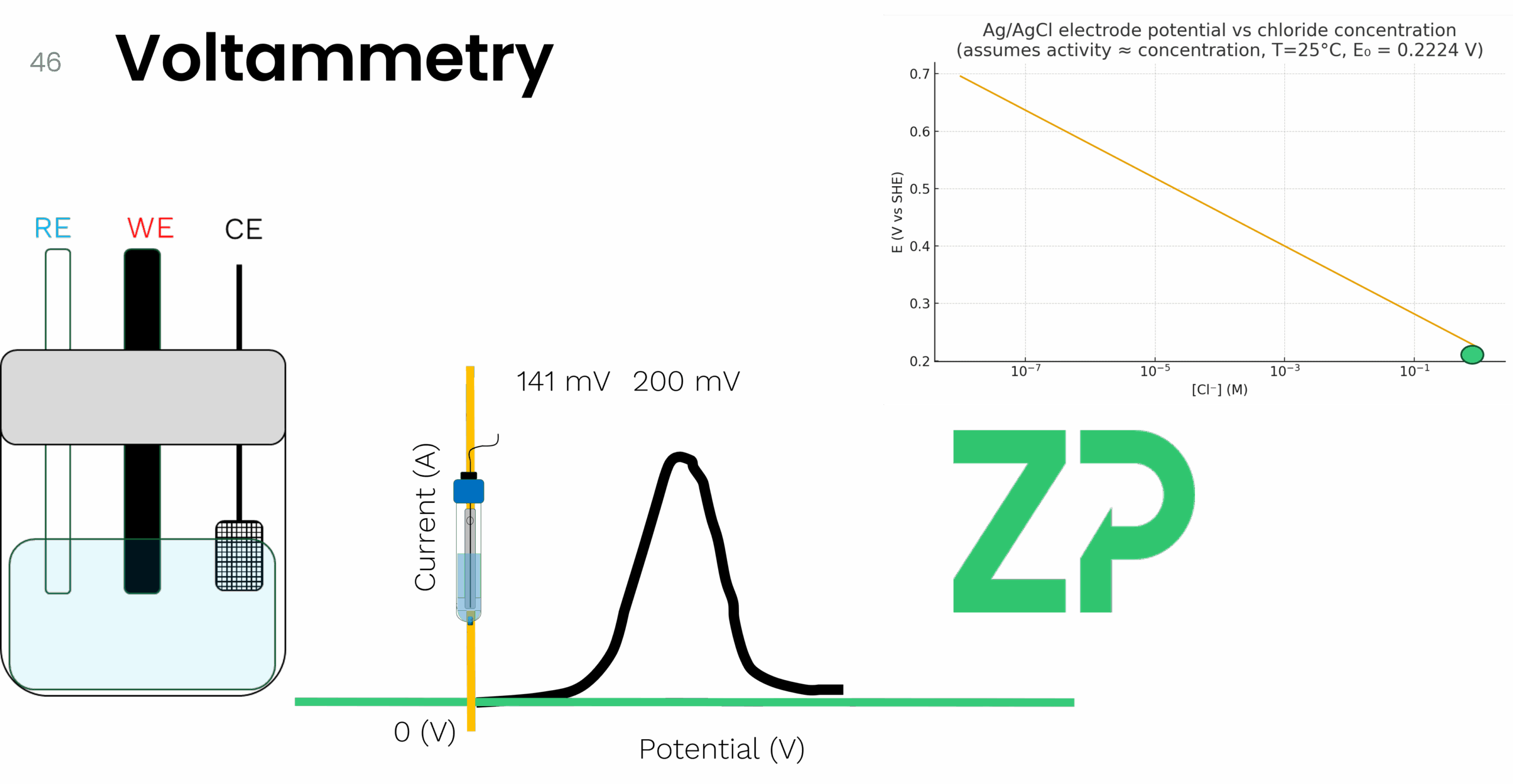
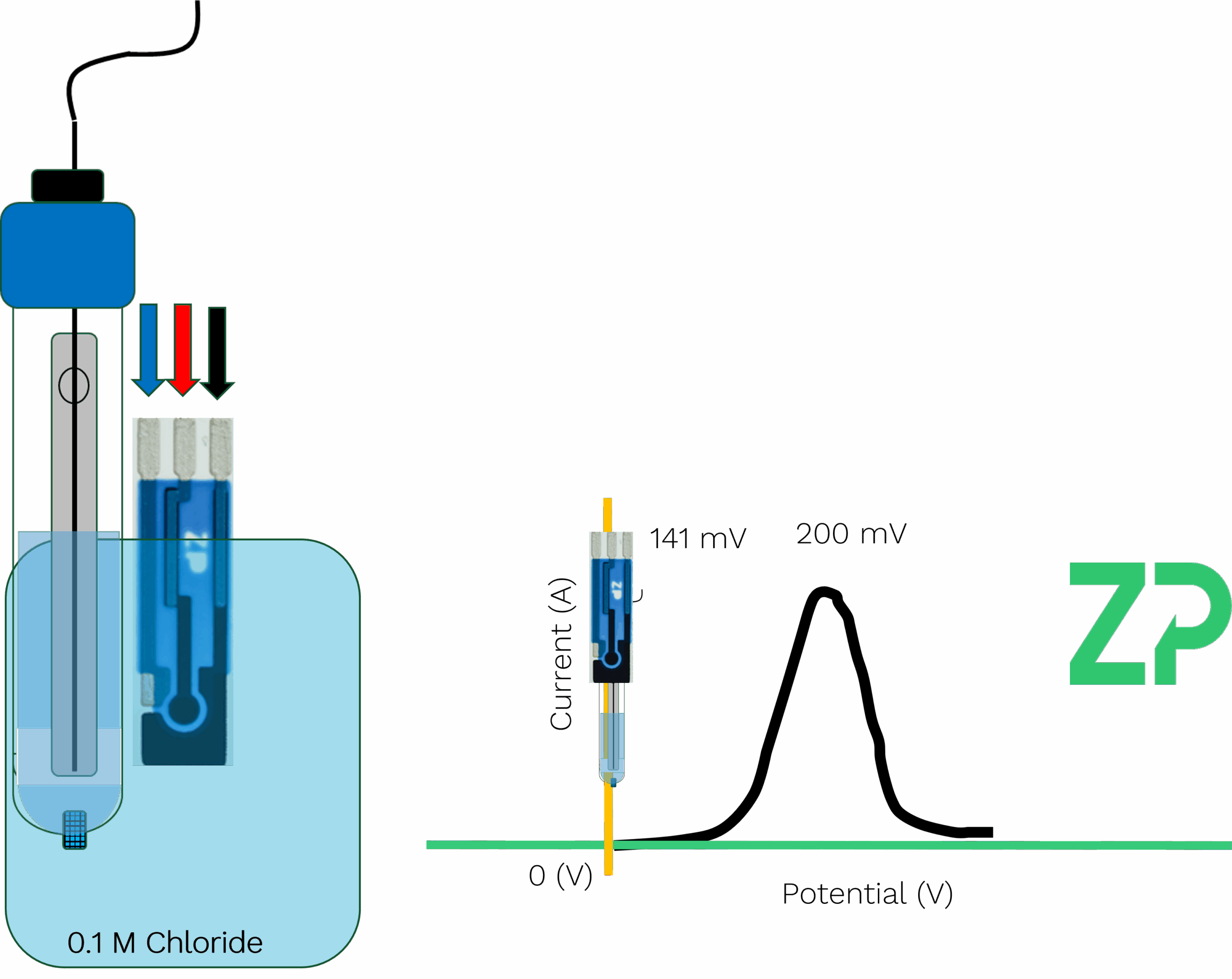
Where: • is the standard potential (approximately +0.222 V vs. SHE for Ag/AgCl in 1.0 M KCl) • is the chloride ion concentration Because the chloride concentration is fixed and isolated behind the porous junction, the reference potential remains stable and independent of the external solution. 🧪 What Is a Pseudo Reference Electrode? Now let’s contrast that with pseudo reference electrodes—commonly found on screen-printed electrodes (SPEs). These are often just a silver or carbon track printed onto the substrate, sometimes chloridized, but crucially, they lack the internal electrolyte reservoir and frit that stabilize the potential. For example, on a ZP501 carbon SPE, you’ll see a small gray area—this is the pseudo reference electrode. It’s compact, cost-effective, and ideal for point-of-need testing and wearable applications. But it comes with a caveat: its potential is not fixed. It depends on the surrounding solution, particularly the chloride ion concentration. ⚠️ Why Chloride Concentration Matters Let’s run a thought experiment. Imagine you're using a true Ag/AgCl reference electrode with 1.0 M KCl. You perform voltammetry on a molecule and observe an oxidation peak at +200 mV. Now, if you dilute the KCl inside the reference electrode to 0.1 M, the reference potential shifts by approximately 59 mV (due to the logarithmic relationship in the Nernst equation). That same oxidation peak would now appear at +141 mV. This shift can lead to misinterpretation of results if you're unaware of the change in reference potential. Now consider someone else using the same working and counter electrodes but relying on the onboard pseudo reference electrode of the SPE. If their test solution has a different chloride concentration—say, 0.1 M instead of 1.0 M—their observed peak potential will also shift by ~59 mV. This variability can be problematic if not accounted for. 🔬 Why Pseudo Reference Electrodes Are Still Useful Despite their sensitivity to solution composition, pseudo reference electrodes are widely used because: • They’re integrated into compact, disposable formats like SPEs • They enable portable, point-of-care, and wearable applications • They offer consistent results when used with standardized conditions In most practical applications, users maintain a consistent chloride concentration across experiments, ensuring repeatability. But if you change the chloride concentration—even unintentionally—expect your potentials to shift. 🧾 Other Types of Reference Electrodes While Ag/AgCl is the most common, here are a few others worth mentioning:
🧠 Final Thoughts True reference electrodes offer stability and precision, ideal for controlled lab environments. Pseudo reference electrodes, while less stable, are indispensable for modern, real-world electrochemical applications—especially when size, cost, and portability matter. Just remember: chloride concentration matters. Keep it consistent, and you’ll get consistent results. If you have any questions about electrochemistry or reference electrodes, feel free to reach out to us at Zimmer and Peacock. We’re always happy to help. Thanks for reading—and happy experimenting!